 
Because each element has a unique number of electrons, an atom will release energy in a pattern unique to its elemental identity and thus will absorb photons in a correspondingly unique pattern. 
In optical spectroscopy, energy absorbed to move an electron to a higher energy level or the energy emitted as the electron moves to a slower energy level is absorbed or emitted in the form of photons. Electrons may move between orbitals, but in doing so they must absorb or emit energy equal to the energy difference of their atoms specific quantized orbital energy levels. 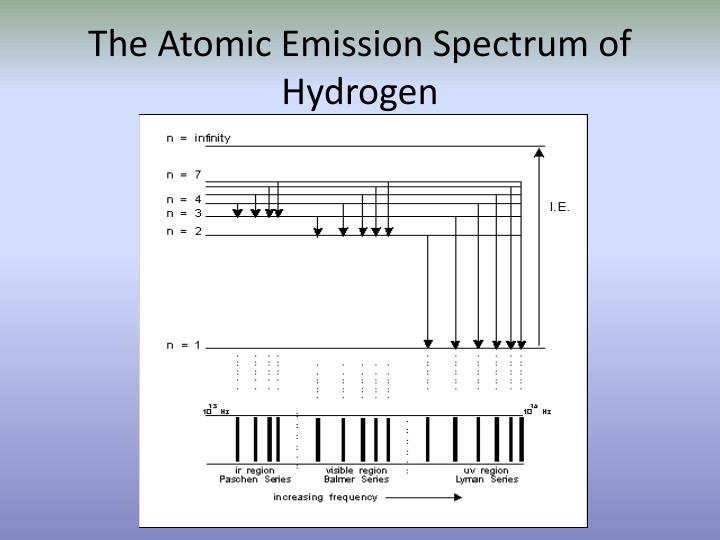
In emission spectra, bright lines will show up corresponding to the difference between energy levels of the elements where in an absorption spectrum, the lines will be dark since every element has unique energy levels, the spectra can help identify elements in a sample.Įlectrons exist in energy levels within an atom atomic orbitals are quantized, meaning they exist as defined values instead being continuous. Because each element has a different number of protons in the nucleus, the energy level of each element is unique. Since unique elements have characteristic spectra, atomic spectroscopy, specifically the electromagnetic spectrum or mass spectrum, is applied for determination of elemental compositions. Every element has a characteristic arrangement of electron orbitals.Ītomic spectroscopy is the study of the electromagnetic radiation absorbed and emitted by atoms. Due to the nature of quantum physics electrons can absorb and emit only specific discrete quantum physics, electrons can absorb and emit only specific discrete energies. Hint: Atomic emission spectra represent the electrons in an atom returning lower energy levels. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
